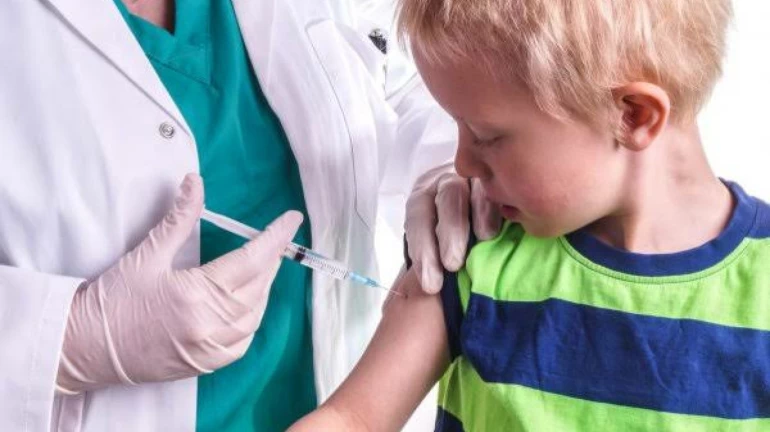
The Director of All India Institute Of Medical Sciences (AIIMS), Delhi, Dr Randeep Guleria has revealed that vaccinations for children could begin by September this year.
Guleria, who is also a member of the Union Government’s COVID-19 Task Force, said that Covaxin data from Phase 2 and 3 trials among kids will be ready by September, adding that approval is expected at around the same time.
Read - ‘Calf Serum’ In Covaxin: Union Health Ministry Issues Clarification Over Social Media Claims
Speaking to India Today, Guleria further said that the Pfizer-BioNTech coronavirus vaccine could also be an option for children if it receives the requisite approvals by then.
Notably, the Drugs Controller General of India (DCGI) on May 12 granted Bharat Biotech permissions to begin phase 2 and phase 3 trials on kids aged 2 and over.
Addressing concerns about the reopening of schools, Dr Guleria mentioned that officials should consider opening schools while making sure they don’t turn into super-spreader locations. He further said that open-air schools could be a way to avoid infections in a classroom environment, as long as the weather/climate permits.
The COVID-19 Subject Expert Committee (SEC) of the Central Drugs Standard Control Organisation (CDSCO) has accepted the phase 3 trial data submitted by Bharat Biotech for Covaxin.
As per the data submitted by Bharat Biotech, Covaxin exhibited 77.8 per cent efficacy in its trial that used 25,800 subjects. Reports suggest that the SEC recommendations have been sent to DCGI, which will now further review the data.
Also read - Covaxin, Covishield Or Sputnik V? AIIMS Director Randeep Guleria On Which Vaccine Is Most EffectiveIndia Today





