A large-scale cervical cancer prevention campaign has been initiated in Maharashtra, where nearly 9.84 lakh 14-year-old girls are set to be vaccinated over a three-month period. The drive has marked the state’s formal participation in the nationwide Human Papillomavirus (HPV) immunisation programme, following which the vaccine is to be incorporated into routine immunisation services.
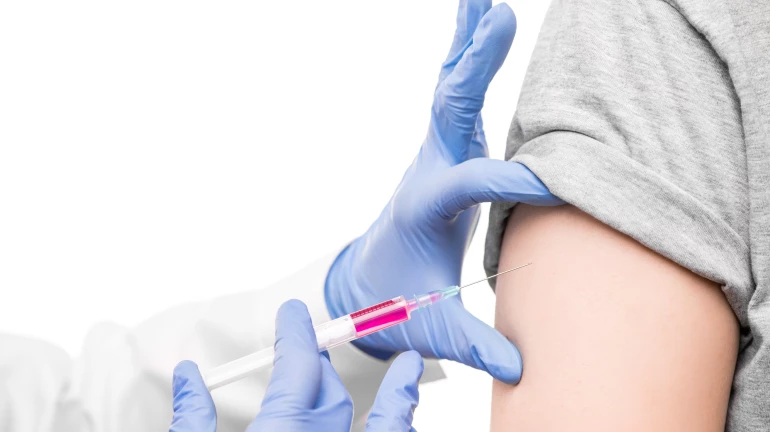
Prior to the national launch by the Prime Minister in Ajmer, Rajasthan, approximately 9,47,380 doses of the quadrivalent vaccine Gardasil-4 were procured by the state. Protection against four HPV strains has been ensured through this vaccine, including types 16 and 18, which have been linked to nearly 70 per cent of cervical cancer cases worldwide. The doses have been stored at the state vaccine depot in Pune under strict cold chain conditions between +2°C and +8°C. Vaccine Vial Monitors have been deployed to ensure that potency has been maintained and that exposure to excessive heat has not occurred.
Eligibility has been limited to girls who have completed 14 years of age but have not yet turned 15. A single 0.5 ml dose is to be administered intramuscularly in the left upper arm. For identification purposes, a mark is to be applied on the left little fingernail. Participation has been declared voluntary, and parental consent has been made mandatory.
Vaccination services are to be delivered through Primary Health Centres, rural hospitals, district and regional hospitals, Municipal Corporation facilities and Government Medical Colleges across the state. Digital registration and certification processes have been facilitated through the government’s U-WIN portal, where OTP-based consent can be provided by parents. Physical consent forms are to be accepted in areas facing limited internet connectivity.
Extensive preparations have been reported in advance of the rollout. Training workshops were conducted, and thousands of officers and frontline health workers—including ASHA and Anganwadi personnel—were prepared for implementation. Coordination has been established with departments responsible for school education, women and child development and tribal welfare to ensure broad outreach.
Mild side effects such as fever, headache or swelling at the injection site have been anticipated, with medical facilities arranged at each vaccination centre. The vaccine has not been recommended for girls outside the specified age bracket, those who are pregnant, severely ill, allergic to yeast or previously vaccinated with other HPV vaccines. Parents have been advised to ensure that girls attend vaccination sessions after meals to reduce the risk of fainting.
Upon completion of the three-month campaign, HPV vaccination is to be integrated into routine immunisation days throughout the year. The initiative has been promoted under the slogan emphasising protection for daughters’ futures, and a ceremonial launch in Maharashtra has been scheduled at B.Y.L. Nair Hospital in Mumbai, where vaccinations are to be administered in the presence of state health officials.





